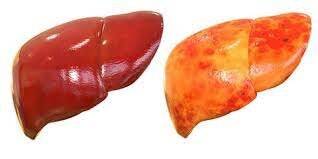
Celiac disease (CD) is an immune-mediated disease that requires a lifelong gluten-free diet (GFD). Although the gluten-free diet is a safe therapeutic option, it can lead to adverse metabolic alterations and increase cardiovascular risk. Why? In recent years, an increase in the risk of non-alcoholic fatty liver disease (NAFLD) has emerged in both children and adults with CD on a GFD, compared to the general population. NAFLD is associated with an increased risk of liver cirrhosis and primary liver cancer. Alarmingly, recent evidence has suggested that NAFLD also increases long-term mortality from cancer, liver diseases, and cardiovascular diseases in children and young adults. Since most celiac patients start a gluten-free diet at a young age, further studies are needed to understand this phenomenon and reduce the risk of liver-related and all-cause morbidity and mortality. The pathogenic link between the gluten-free diet and “fatty liver,” however, has yet to be fully clarified. Persistent alterations of the gut-liver axis and an unfavorable composition of the gluten-free diet have been proposed as potential etiological factors. While reliable and non-invasive assessments of altered intestinal permeability can be difficult in daily clinical practice, it is possible to analyze the intake of packaged gluten-free foods (PGFF). In this work https://www.mdpi.com/2072-6643/14/14/2942/htm the nutritional profile of celiac patients on a gluten-free diet was evaluated through detailed nutritional interviews. Although this approach is particularly suitable in clinical studies, it can be time-consuming and may be difficult to apply in real-life clinical practice, where physicians have to examine a large number of patients in a relatively short period. As a result, simple tools are needed to help physicians identify celiac patients at high risk of NAFLD and therefore in need of tailored nutritional intervention. The main objective of the study was to verify whether dietary habits, measured according to a specific analysis, were independently associated with non-alcoholic fatty liver disease in celiac patients adhering to a gluten-free diet. The secondary objective was to verify whether specific categories of packaged gluten-free foods (PGFF) were associated with a different risk of NAFLD. NAFLD in CD patients adhering to a gluten-free diet has been recently described, but its determinants remain unclear. NAFLD has also been reported in a considerable portion of lean subjects and independently of typical metabolic risk factors. Ultimately, it has been shown that: (1) a relative increase in the consumption of packaged gluten-free foods was significantly correlated with non-alcoholic fatty liver disease; and (2) not all packaged gluten-free foods were associated in the same way with the steatosis in question; rather, specific consumption patterns were particularly at risk. In general, it is known that packaged gluten-free foods have unfavorable nutritional characteristics compared to their gluten-containing counterparts, particularly in terms of lipid and carbohydrate content. Whether packaged gluten-free food subclasses are "less healthy" remains a matter of debate. In their original survey on Austrian packaged gluten-free foods, Missbach et al. did not find significant nutritional differences in bread and snacks. However, other studies conducted with similar methodologies showed that Canadian, Norwegian, Slovenian, Moroccan, and Brazilian gluten-free breads had a higher carbohydrate and saturated fat content compared to gluten-containing breads. Similar differences have also been reported for sweet ready-to-eat foods, another category particularly associated with NAFLD in the study. Conversely, nutritional differences in other categories were less relevant. So far, no study has verified whether these nutritional differences also translate into actual metabolic alterations. Therefore, we have provided new information that could prove useful for improving the primary and secondary prevention of NAFLD in celiac patients. The results have substantially confirmed the hypothesis that attributes liver disease mainly to the composition of packaged gluten-free foods. Two different mechanisms may contribute to steatogenesis. First, a greater amount of carbohydrates and fats in the small intestine may lead to a greater amount of these nutrients reaching the liver through the portal flow. Second, short-chain fatty acids could interact with the gut microbiota, leading to dysregulated production of acetate and propionate (two regulators of de novo lipogenesis in the liver). These data also provide insights that could be useful in the nutritional management of celiac patients, particularly for those at risk of steatosis. Three patterns of nutritional behaviors should be well monitored. The first is the purchase/consumption of large quantities of packaged gluten-free foods per month. The second is the purchase/consumption of a large variety of different packaged gluten-free foods during the last week, which means a preference for such products and, finally, a preference for specific packaged gluten-free foods that are particularly linked to NAFLD (in the study: bread and bakery products, both sweet and savory). This information is relevant for providing nutritional advice and suggestions on new dietary approaches for celiac patients to avoid metabolic disorders, including NAFLD. For example, patients should be encouraged to increase their consumption of olive oil, legumes, unrefined grains, fruits and vegetables, and introduce pseudocereals as a source of complex carbohydrates, proteins, fibers, fatty acids, vitamins, and minerals. The information was obtained through a simple, reproducible, and time-saving questionnaire, which can be used in clinical practice to create tailored approaches for the patient and in future studies to validate the results in different geographical areas.


©

Celiachia e steatosi epatica non alcolica
Celiachia e steatosi epatica non alcolica
2022-08-23 11:16
2022-08-23 11:16
Romina Giuliani
Romina Giuliani //www.microbiotaedintorni.it/favicon.png
celiachia, nutrizione, microbiotaedintorni, celiachia, steatosi-epatica-non-alcolica,

©