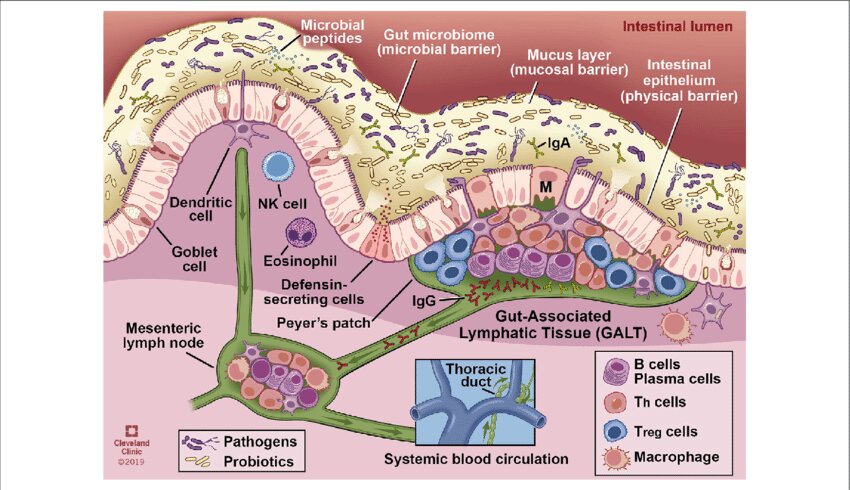
The gut-associated lymphoid tissue (GALT) is the largest immune organ in the human body. It is a well-developed component of the intestinal mucosal immune system, involved in protecting the host from pathogenic antigens and in the maturation of the postnatal immune system. The intestinal microbiota indirectly mediates resistance to infections by stimulating the innate immune response. Differences in the composition of microbial communities are associated with the host between states of health and disease. Genetic predisposition underlies autoimmune diabetic pathology but must be triggered to induce the autoimmune reaction. The best hypothesis so far is that the trigger occurs in the gut. The intestinal microbiota remains in direct contact with the external environment and provides the ideal site for the immune response. In fact, below the intestinal mucosa is the GALT, which is the lymphoid tissue associated with the intestine and contains a vast population of various types of immune cells, consisting of Peyer’s Patches (PP), mesenteric lymph nodes (MLN) and permanent or transient lymphoid follicles. The PP are oval or round in shape and are found in the lowest part of the small intestine, especially in the jejunum and distal ileum and to a lesser extent in the duodenum. The PP are made up of APC (DC), mononuclear cells (macrophages, T cells, B cells). B cells are found in the follicular germinal centers, while T cells are distributed in the area between the follicles. All lymphoid follicles are covered by follicle-associated epithelium (FAE), in which there is a lower number of goblet cells that produce mucus and Microfold cells (M cells). The latter are associated with the absorption and transport of antigens from the lumen to the APCs. In the FAE, tight junction proteins are expressed that make the epithelium less permeable. The PP play a significant role in the immune response within the intestinal mucosa by monitoring the immune activity of the intestinal lumen in the presence of pathogenic microorganisms. The MLN connect the intestine to the pancreas through the PLN (pancreatic lymph nodes). In particular, the intestinal PP connect to the MLN through the lymphoid circulation. The superior MLN are ventrally connected to the inferior PLN. To date, the understanding of how immune cells migrate from MLN to PLN contributing to the death of β cells is incomplete. It is supposed that alterations of the intestinal microbiota, such as the overgrowth of certain species, or the presence of pathogens, can activate macrophages, T cells, B cells, or dendritic cells in the PP and other sites of the GALT, producing an immune response. These activated immune cells migrate from the GALT to the PLN, carrying out their activity. During the maturation and differentiation of B and T cells, exposure to the intestinal environment, the functions are significantly modulated; for example, exposure to foreign antigens leads to a greater secretion of immunoglobulin A (IgA) and cytotoxic CD T cells in the lamina propria. In children with T1D, the integrity of the intestinal epithelium, including the FAE, is reduced, leading to a leaky gut and allowing antigens to easily penetrate the FAE and stimulate immune cells.


©

GALT e risposta immunitaria nel diabete di tipo 1
GALT e risposta immunitaria nel diabete di tipo 1
2021-11-09 14:09
2021-11-09 14:09
Romina Giuliani
Romina Giuliani //www.microbiotaedintorni.it/favicon.png
microbiota-intestinale, diabete-di-tipo-1, diabete-giovanile, diabete-infantile, diabete-autoimmune, autodistruzione, galt, distruzione, risposta-immunitaria, autoanticorpi,
"Tutte le malattie cominciano nell'intestino.", ci aveva visto bene Ippocrate!

©