Psoriasis is a chronic immune-mediated inflammatory disease that affects about 125 million people worldwide. Several studies regarding the composition of the gut microbiota and its role in the pathogenesis of the disease have recently shown significant alterations among psoriatic patients. Some parameters, such as the Firmicutes/Bacteroidetes ratio, have been developed to distinguish between psoriatic and healthy individuals. The "leaky" gut (permeable) and bacterial translocation are considered by some authors to be a trigger for the onset of the disease, as they promote chronic systemic inflammation. It has also been found that the alterations resemble those of inflammatory bowel diseases, obesity, and some cardiovascular diseases. Dysbiosis of the microbiota, depletion in the production of SCFA (short-chain fatty acids), increased amount of TMAO (trimethylamine-N-oxide), dysregulation of the pathways that influence the balance between lymphocyte populations, all seem to be the most significant results regarding intestinal physiology in psoriatic patients. The pathogenesis of psoriasis is complex and not yet fully understood. The current state of knowledge indicates that genetic predispositions and many immunological and environmental factors (e.g., trauma, infections, drugs, UV and X-rays, chemical and thermal burns, smoking, alcohol consumption, stress) can act as crucial elements, stimulating keratinocytes to begin secreting pro-inflammatory cytokines. This process causes antigen-independent activation of T lymphocytes, which produce TNF-α, IL-1, IL-2, IL-6, IL-8, IL-12, IL-17, IL-23 p19/p40, INF-γ, granulocyte-macrophage colony-stimulating factor (GM-CSF), and vascular endothelial growth factor (VEGF), which affect keratinocytes and blood vessels, leading to abnormal hyperproliferation of keratinocytes, the development of parakeratosis due to the shortening of the keratinocyte maturation process in the epidermis, and abnormal angiogenesis with the formation of twisted and fragile vessels with increased permeability in the areas of skin lesions. Groeg et al. conducted a randomized, double-blind study, including 22 patients with ulcerative colitis, 48 patients with chronic fatigue syndrome, 22 patients with chronic plaque psoriasis, and a control group of 35 healthy people. At baseline, psoriatic patients were identified as having higher levels of serum C-reactive protein (CRP), TNF-α, and IL-6 compared to the control group. The research groups were given a sachet containing 1 × 1010 colony-forming units (CFU) of viable Bifidobacterium infantis 35.264 per day for 6-8 weeks (the duration depended on the type of disease). A significant decrease in plasma levels of CRP, TNF-α but not in IL-6 concentration was observed among psoriatic patients after 8 weeks. The randomized, double-blind, placebo-controlled study by Navarro-Lopez et al. involved 90 patients, who were given a mixture of three probiotic strains in a 1:1:1 ratio: Bifidobacterium longum CECT 7347, B. lactis CECT 8145 and Lactobacillus rhamnosus CECT 8361 with a total of 1 × 10 9 CFU per capsule once a day for 12 weeks. A significant reduction in the PASI score (understood as PASI-75 reduction) was found in 66.7% of patients receiving probiotics and 41.9% of patients receiving placebo after 12 weeks. A 6-month follow-up showed that patients who had received probiotics had a lower risk of relapse of psoriasis, as the administered bacteria modulated their microbiota composition.
The gut microbiota can serve as a potential biomarker of treatment response in some cases of biological treatment. Oral administration of probiotics and fecal microbiota transplantation have been the most reported in bringing health benefits to psoriatic patients. However, the issue of the composition of the psoriatic gut bacteria, its role, and healing potential needs further investigation.


©

Psoriasi e microbiota intestinale: stato dell'arte
Psoriasi e microbiota intestinale: stato dell'arte
2021-10-29 14:40
2021-10-29 14:40
Romina Giuliani
Romina Giuliani //www.microbiotaedintorni.it/favicon.png
microbiota-intestinale, dieta, microbioma-intestinale, malattia-autoimmune, autoimmunita, psoriasi, malattia-immuno-mediata, disbiosi-intestinale, strategie, terapie, probiotici, trapianto-di-microbiota-fecale, prebiotici,
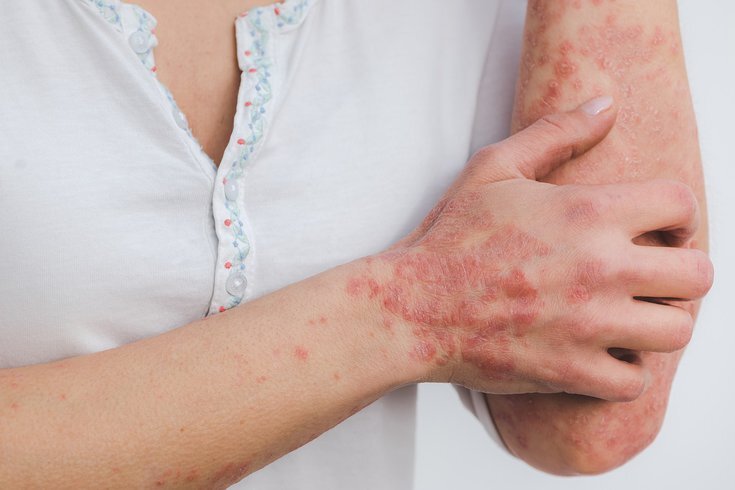
Esiste una relazione tra psoriasi e disbiosi del microbiota intestinale?

©